All Images
News Release 09-185
For Future Superconductors, a Little Bit of Lithium May Do Hydrogen a Lot of Good
Study suggests strategies for converting hydrogen to metal at significantly lower pressures
This material is available primarily for archival purposes. Telephone numbers or other contact information may be out of date; please see current contact information at media contacts.
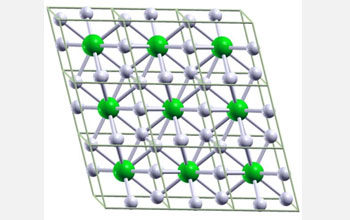
Ball-and-stick image of hypothetical metallic crystal cells composed of one lithium, or Li, atom and six hydrogen, or H, atoms. The lithium-hydrogen compound is predicted to form under approximately 1 million atmospheres, which is one-fourth the amount of pressure required to metalize pure hydrogen. The pressure at sea level is one atmosphere and the pressure at the center of the Earth is around 3.5 million atmospheres. Li atoms are green and H atoms are white.
Credit: Eva Zurek, Department of Chemistry, State University of New York at Buffalo
Download the high-resolution JPG version of the image. (163 KB)
Use your mouse to right-click (Mac users may need to Ctrl-click) the link above and choose the option that will save the file or target to your computer.
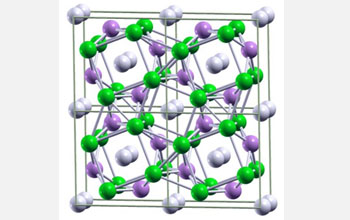
Ball-and-stick image of predicted metallic lithium-hydrogen crystal cells made of one lithium, or Li, atom and two hydrogen, or H, atoms. Li atoms are green, hydrogen pairs are white, and negatively charged H atoms are mauve.
Credit: Eva Zurek, Department of Chemistry, State University of New York at Buffalo
Download the high-resolution JPG version of the image. (182 KB)
Use your mouse to right-click (Mac users may need to Ctrl-click) the link above and choose the option that will save the file or target to your computer.


