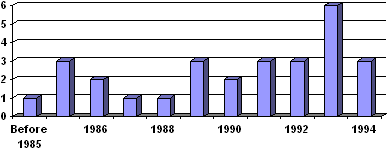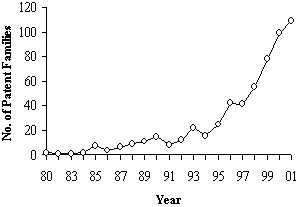|
The Emergence of Tissue Engineering as a Research Field |
|
Back to Table of Contents View PDF of this document (82 Kb) 6.0 Activities in the Corporate SectorTissue engineering remains, in many respects, an eclectic mix of topical foci and research styles, with work of an ad hoc or "Edisonian" character continuing to play a strong role especially in the corporate sector. Overall, the corporate sector is recognized as having played a notable role in the development of this unique field, mostly due to the high level of corporate R&D funding injected into the field as compared to the relatively small influx of funds from the federal government. As many of our interviewees note, TE has traditionally been considered a high-risk investment. As a result, few agencies or program officers in the government—NSF's Division of Bioengineering and Environmental Systems (BES) being a notable exception—were willing to provide funds for such new and creative research. Many interested in pursuing research in TE were often forced to find alternate means of funding, such as by bootstrapping funds from other grants, patent revenue or clinical department revenues, or by bringing their ideas to the private sector. Thus, much of the work in tissue engineering in the corporate sector is a result of a direct transfer of academic work to industry in a rush to bring viable products to market. However, because corporate R&D has traditionally focused on the creation of proprietary intellectual content and less on the solution of broader challenges in science or engineering, knowledge transfer from industry back to academia has been limited. The WTEC110 report on tissue engineering provides a brief overview to the corporate state of affairs:
The types of TE firms can be divided into four major categories: (1) structural applications, (2) metabolic applications, (3) cellular applications, and (4) other enabling technologies (some firms may actually fall into multiple categories, but are classified here by their primary focus areas). In Chapter 4, we provided information on some of the major tissue engineered products and the companies which produce them. In this section, we examine 28 firms that started before 1994, in order to understand the character and influence of such firms during the period when tissue engineering was just beginning to emerge. Figure 6.1 lists the years the companies began. Since our goal was to investigate private sector activity in the early years of tissue engineering, we limited our exploration to companies that started before 1994. Table 6.1 on the next several pages lists US-based companies in the early days of the field, and (consistent with the theme of this report) examines their origins. Table 6. 1: A Sample of Tissue Engineering Companies and their Origins |
| Name | Year Started | Founded By | Founder Affiliation (if known) | Origin/Institutional Links |
|---|---|---|---|---|
| Interpore Cross International, Inc. | 1975 | Edwin Shors co-founder, Edward Funk, Ingeborg Funk, | Company founded on patent held by inventor at Pennsylvania State University | The company originated as a product of the ideas of 3 people from Penn State University and Penn State College in early to mid 1970s. Eugene White, a material scientist from Penn was interested in uses of a scanning electron microscope, he had a colleague (now deceased) at the Department of Marine Geology who was interested in corals as indicators of geological events. White's nephew Rodney White (now chief of vascular surgery at UCLA) was looking for a job. They collectively came up with the notion of making materials with inteconnected porosity (similar to corals) which may be optimal for connective tissue growth bone growth. They made a variety of implants and realized the medical value of the experiments (particularly in the area of cardivasicular applications paticularly prostheses). The company was eventually founded on that principle. The concept was patented with Penn State holding the patent. The group called their work tissue gardening. |
| BioHybrid Technologies | 1985 | Founded by Bill Chick and John L. Hayes | Harvard Medical School | Bill Chick had set up a diabetes unit at U Mass Worcester, and was interested in developing artificial pancreas using insulin-producing microreactors; found that expensive to fund with grants; started to look - together with John Hayes - for funding from industry. First 8 years of company supported by WR Grace. In 1992, WR Grace and Biohybrid parted, and Grace bank-rolled Cerce (put artifical pancreas project on ice). Biohybrid went on its own with microencapsulation technology; funded by NIST ATP 4 year grants, JDF grant; NSF SBIR grant; no university affiliations; had advisory boards that have academics and academic consultants |
| Celox Laboratories, Inc. (merged with Protide in 2001) | 1985 | Milo R. Polovina has been President, Chief Executive Officer, Treasurer, and Secretary of the Company and has served as a Director since 1985. | Company started to research, develop, manufacture and market cell biology products that are used in the propagation of cells derived from mammals, including humans and other species; Global marketing agreement with ICN Pharmaceuticals; signed an option agreement with the University of Minnesota Office of Patents and Technology Marketing for an infusible grade solution for non-cryopreserved human hematopoietic stem cells. This option agreement allowed Protide to have the exclusive right to evaluate the technology and possibly commence negotiations with the University for worldwide commercialization. | |
| Creative BioMolecules, Inc.(now Curis Inc after merger with Ontogeny and Reprogenesis) | 1985 | Charles Cohen, Fred Craves, Roberto Crea | Jay Vacanti and Bob Langer of MIT thought about a way of using polymers and cells to make new tissues, and that led to Neomorphics, which subsequently merged with Advanced Tissue Sciences. A portion of these patents got licensed to Reprogenesis, which is now part of Curis; Reprogenesis also had start-up support from Pfizer. Curis has collaborations with Stryker Corporation, Biogen, Inc. | |
| LifeCell Corporation | 1986 | Steve Livesay | University of Texas, Austin | University of Texas, Austin; More than 15 years ago, in a laboratory at the University of Texas, Dr Stephen Livesey, MD, PhD, and his colleagues were studying the formation of different structures of ice. Their goal was to develop a method of freeze-drying biological tissues and cells without damaging their structural or biochemical integrity. What they invented was the first of the patented preservation technologies which form the basis of LifeCell's technology today. Livesay transferred this cryopreservation technology from the University into the start-up. Now company has links with Boston Scientific; has received many NIH SBIR grants and DOD ATP grants |
| Organogenesis, Inc. | 1986 | Eugene Bell and two postdoctoral students (Christian Weinberg and unknown) | MIT | Product marketed by Novartis Pharma AG |
| Advanced Tissue Sciences, Inc. | 1987 | Gail Naughton | New York University | Company based on Naughton patent; Jay Vacanti and Bob Langer thought about a way of using polymers and cells to make new tissues, and that led to Neomorphics, which subsequently merged with Advanced Tissue Sciences. Smith and Nephew has provided support for cartilage and skin development since 1994. |
| Protein Polymer Technologies | 1988 | No information | ||
| Cytotherapeutics (Stem Cell Inc.) | 1989 | David Scharp, Paul Lacy, Michael Lysaght (Baxter) | Michael J. Lysaght, Associate Professor of Artificial Organs (Research) at Brown University. | Brown University, Other partners include: California Institute of Technology (Technology Licensing Agreement), Oregon Health Science University, Scripps Institute. Company split into successor companies: Stem Cells Inc. (Palo Alto, CA), Neurotech Corp. (Lincoln, RI), and Modex Therapeutics SA (Lausunne Switzerland). In 1989, Lysaght left Baxter to help start CytoTherapeutics. He served as Vice President and chief technical executive at CytoTherapeutics from 1989 through 1994. |
| ETEX Corporation | 1989 | D. Duke Lee | Lee is the Chairman, Chief Scientific Officer and the scientific founder of ETEX. He is on the faculty of Harvard Medical School and also serves as Director of the Harvard/MIT Biomaterials Training Program. | Agreement with several pharma companies: Biomet Merck as the exclusive distributor of Biobon in Europe and Latin America; DePuy Orthopaedics, Inc. a Johnson & Johnson company, for distribution of ETEX alpha-BSM ®, Bone Substitute Material, for orthopaedic indications, and joint research and development of future products; Sofamor Danek Division of Medtronic, Inc, to jointly develop products for spinal applications. |
| Integra LifeSciences Corporation | 1989 | Richard Caruso | No further information | |
| REGEN Biologics, Inc. | 1990 | Kevin Robert Stone | Privately funded by Sulzer Medica, Sanderling, Sequoia Capital, and Allen & Company | |
| Synthecon, Inc. | 1990 | Charles Anderson | NASA contractor company | Co-founders C.D. "Andy" Anderson and Ray Schwarz worked for a NASA medical services contract company. As members of a Space Bioreactor Project Team, their charge was to develop a bioreactor that would enable scientists to study the effects of space on human tissue and help them understand why astronauts suffered bone and muscle loss in orbit. With assistance from NASA astronaut David Wolf, M.D., and medical contractor Tinh Trinh, Schwarz invented a fluid-filled rotary wall vessel (RWV) bioreactor that enabled NASA scientists to successfully grow, maintain and study three-dimensional human tissue in space for extended periods of time. |
| Aastrom Biosciences, Inc. | 1991 | Bernhard Palsson, Michael Clarke, Stephen Emerson | Hematologists at the U Mich School of Medicine; Palsson is now a Professor of Bioengineering and Adjunct Professor of Medicine at the University of California, San Diego | A 1989 research agreement with the University of Michigan, and licensing patents based on University-conducted research (relating to the ex vivo production of human cells), collaborations also with University of Colorado (to insert cell-destruction genes into AIDS patients' stem cells, which then would be expanded in the bioreactor and transplanted back into the patient). First approached by large pharmaceutical firm but then given away to venture capital firm. |
| Layton BioScience, Inc. | 1991 | James Eberwine, Virginia M.-Y. Lee, and John Q. Trojanowski | The three founding scientists were all neuroscientists at University of Pennsylvania | The technologies initially being developed were discovered in the laboratories of the company's founding scientists and licensed from Stanford University (Stanford) and the University of Pennsylvania (Penn). Cooperation, Licensing and/or Other Agreements with: Incyte Genomics, Inc, Merck & Co, PerkinElmer Life Sciences, Stanford University, Stratagene Corp, University of Florida (U. S.), University of Pennsylvania, and University of Texas |
| ORTEC INTERNATIONAL | 1991 | Steven Katz, Ron Lipstein, Mark Eisenberg, Alain M Klapholz | Ortec�s technology is based on the technology developed by Mark Eisenberg, an Australian physician | |
| Orthovita, Inc. | 1992 | Paul Ducheyne | University of Pennsylvania, Bioengineering and Orthopaedic Surgery Research | Ducheyne is currently Chairman Emeritus and Director of Orthovita. Since 1997, Dr. Ducheyne has been the Director of the Center for Bioactive Materials and Tissue Engineering at the University of Pennsylvania |
| TEI Biosciences | 1992 | Eugene Bell | MIT | No information provided |
| Prizm Pharmaceuticals, Inc., (merged with Matrigen, Inc. in 1998 to form Selective Genetics) | 1992 | Andrew Baird, Samuel Ward Cassells, III, (Prism) Steven Goldstein, Robert Levy (Matrigen) | Andrew Baird (then at Scripps), Ward Cassells, University of Texas) started Prism. Steven Goldstein (University of Michigan) and Robert Levy, (Childrens hospital) started Matrigen | Matrigen licensed technology from U Michigan; Prizm licensed technology from Scripps Clinic; Prizm started by private venture firms DOMAIN ASSOCIATES and OXFORD BIOSCIENCE PARTNERS they started it as a company to stop cell proliferation - toxin conjugated to a growth factor. Idea was that the body will internalize growth factor, body will stop cell proliferating. Was not feasible. Method worked only with toxicity - had to abandon technology. In 1998, met up with another company simulating cell growth (matrigen) resulting company Selective Genetics to explore tissue repair and regeneration |
| Fibrogen | 1993 | Thomas Neff | Neff was an investment banker with both PaineWebber and Lazard Freres & Co., and for many years he has followed commercial and scientific developments relating to molecular biology | A 1996 collaborative agreement with the University of Pittsburgh Medical Center re: biotech liver system. |
| Guilford Pharmaceuticals | 1993 | Scios Nova Inc., and Solomon Snyder and Craig Smith | Bob Langer of MIT conceived of degradable polyanhydride systems, and licensed that originally to a company called Nova Pharmaceuticals. They merged with Scios, and then they spun off Guilford. | |
| Osiris Therapeutics, Inc. | 1993 | Arnold Kaplan, Steven Hanesworth (cell biologist), Victor Goldberg (orthopedic surgeon), James Burns, VC and Unnamed Businessman | Kaplan, Hanesworth, Case Western Reserve University, had collaborations with Goldberg at the University Hospital | Kaplan worked with Burns in 1992 to get company started, founders stayed involved till 1997-98 through their participation on a scientific review board, the company also continued collaboration with the founders through about 1995 when it moved to Baltimore. Current CSO Marshak also holds an appointment as Adjunct Associate Professor of Oncology and of Molecular Biology & Genetics at the Johns Hopkins University School of Medicine. Marshak has served on several NIH Study Sections, the Medical and Scientific Advisory Board of the Dystonia Medical Research Foundation, and the Editorial Board of the Journal of Biological Chemistry. |
| OsteoBiologics, Inc. | 1993 | cofounders faculty at Univeristy of Texas Heath Science Center (Barbara Boyan, Athanasiou) one business founder | licenisng tech develoepd own, have R&D agreements with other cooperations, company, collaborators at Universities | The technologies initially being developed were discovered in the laboratories of the company's founding scientists and licensed from Stanford University and the University of Pennsylvania. Began by licensing technology developed at the University of Texas Health Science Center |
| Therics | 1993 | Brad Vale (J&J development corporation), Walter Flamenbaum; serial founder of companies starting at NYC's Mt. Sinai School of Medicine) | Licensed technology developed at MIT (work of Linda Griiffith, Michael Cima, and Ely Sachs) | Boston University (U. S.) (Technology Licensing Agreement), Massachusetts Institute of Technology (U. S.) (Technology Licensing Agreement) |
| Ximerex | 1993 | William Beschorner. | Founder was at Johns Hopkins Hospital came up with the idea, developed it, filed patent and started company, | Has links with the University of Nebraska, started out at Johns Hopkins (1993-95), private lab, in 1997 moved to Omaha |
| Islet Technology | 1994 | Scott Wiele | Founder's daughter diagnosed with Type1 diabetes; father wanted to know if cure (rather than management) was possible; starterd searching and found a program at UC Davis. Kent Cochrum had technology for encapsulating islets from transplants and was funded by another compay, but asked Wiele to help him raise more money. Wiele licensed technology. Company has worked closely with U Minn and Diabetes Institute (Barnard Hering) via licensing arrangment | |
| MultiCell Associates | 1994 | Galletti and Jauregui with Jayanta Roy Chawdhury, and Alfred Vasconcellos | Albert Einstein School of Medicine | Wholly owned subsidiary (2001) of Exten CA |
| Orquest, Inc. | 1994 | strategic partners are other firms http://www.orquest.com/wt/sec/strat_partners |
|
Figure 6.1: New Tissue Engineering Companies by Year (1994 and earlier) The focus areas of these firms were as follows:
The companies listed above were clustered in specific parts of the country. Of the 28 companies examined, eight were in California, five in Massachusetts, three each in New Jersey and Texas, and two each in Minnesota and Rhode Island. Michigan, Delaware, Maryland, Nebraska, and New York, all had one company each. Their locations parallel the locations of many of the major research centers in TE, which supports the hypothesis that many of these companies have close ties to academia—at least at the start-up phase. In fact, sixteen of the 28 companies had at least one founder in academia. Only five of the 28 companies had co-founders that were neither currently nor formerly academics (i.e. were based in business or venture capital)111. Twenty one companies licensed or transferred intellectual property from academia (without necessarily having an academic co-founder). Two did not (Synthecon—NASA contractor, and Ortec, Australian physician)112. Table 6.1 above summarizes the origins of these companies. As Table 6.2 below shows, the universities involved in start-ups were: Table 6.2: University Start-ups in Tissue Engineering
Of the 28 companies, 12 are public and 14 continue to be privately held (no information was available on the status of the remaining two firms). Financial support for these companies came from a variety of sources as shown in Table 6.3 below. Table 6.3: Financial Support of Corporate Start-ups
All of the firms listed here were still in existence through the close of 2001 in one form or another (many have merged with others to form new companies, but none have disappeared altogether) and collectively employ (in 2001) about 2000 people. The overall number of firms and, consequently, employees in tissue engineering, has steadily increased since 1994113. Little data is available on the specific training of the researchers employed by private companies active in tissue engineering, and it is very difficult to paint a precise picture of the characteristics of the individuals that make up this group. However, the fragmentary data available to the study team suggest that few of the scientists and engineers employed by companies active in tissue engineering possess formal credentials in the field. Rather, companies seek individuals who have broadly-applicable technical knowledge and skills relevant to the research and development tasks they face, and assign these individuals to specific projects as required; employees in the corporate sector may enter or leave tissue engineering as an activity quite freely. Flexibility is especially important in that no start-up company in the field that has attempted to develop cell-based products has yet established a successful business, and the ability to redirect staff to the most promising lines of work - often away from initially ambitious product concepts to simpler ones with more likely prospects of reaching the market in the near term—can be critical for corporate survival. With the caveat that our data are limited, we encountered no evidence that movement of newly-trained junior researchers from academia to industry has been an important mechanism of technology transfer in tissue engineering. At the senior level, although a handful of investigators have left academia to assume full-time roles leading start-up companies, more frequently senior academics in the field will serve as advisors or board members for companies licensed to develop technologies or product concepts that emerge from their laboratories. Review of Patent Activity: Domestic and International As an alternative means of understanding the origins of tissue engineering and progress made in the field, we also examined patenting in the field over the past twenty or so years. The full patent analysis is included as Appendix 5 at the end of this report.
As the adjoining figure shows, the earliest patenting activity occurred in the mid-to-late 80's with a more dramatic increase in the 1990s; consistent with the overall growth in awareness of the field. As the Figure shows, patenting in tissue engineering has been trending up since 1980 and has not yet peaked. In particular, in the last 5 years, patenting has increased 226% over the previous 5 years, which in turn was an increase of 138% over the prior 5 years. The bulk of this innovation was US-based114, as shown in Figure 6.2. Over seventy percent of the global tissue engineering patents are invented in the US, followed by 18% in Europe (led by Germany and UK) and 6% in Japan. Figure 6.2 - Priority (Inventor) Country of Worldwide Tissue Engineering Patents (1980-2001): N=567 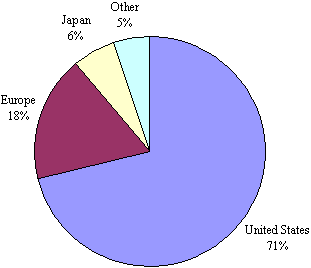 Given that most of the invention is coming from the US, it is not surprising to see that most of the patent assignees are US institutions as well. Figure 6.3 shows all assignees with 4 or more global patent families115. The institutions holding the most highly cited patents are listed in Table 6.4. International institutions are depicted in gray. A list of the 100 most highly cited tissue engineering patent families is also given in Appendix 5 in Table G. The Table shows that the highest relative cited patent family is a 1999 Isotis (a biosurgery company based in Netherlands founded in 1996) patent that has received 11 citations already. Since a typical 1999 patent family has just over 1 citation, this patent is cited 7.5 times as often as expected. The highest overall cited patent family is an Advanced Tissue Science invention "Three-dimensional cell and tissue culture system." This 1990 patent has received 162 citations from later patents, which is almost 6 times the expected number (28.3) for a 1990 tissue engineering patent family. Many of the highly cited patents are coming from the patenting leaders. This is further illustrated in Table 6.4 below, where we see that MIT and Advanced Tissue Sciences have the most highly cited patents by far. Among the most effective patenting companies is Regen Biologics Inc., which has 8 of its 11 patents among the highly cited set.Figure 6.3 - Assignees with 4 or more TE Global Patent Families (1980-2001) 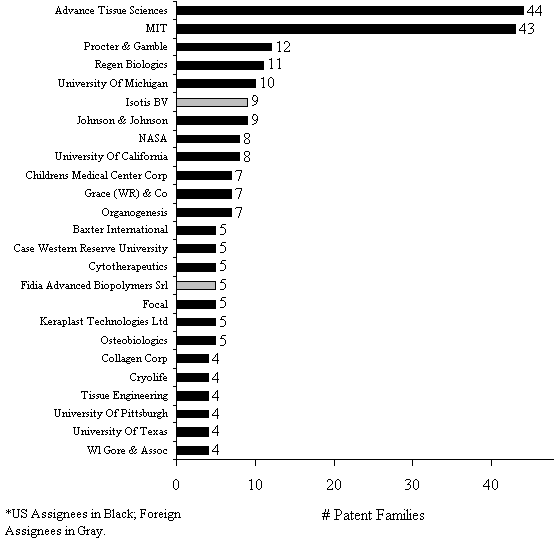 Table 6.4: Share of patents that are highly cited by assignee
In terms of subject matter, of the top 100 most cited patents examined in this analysis, the majority can be classified as cellular, including those which describe methods for cell culture or differentiation and the medium used to support such methods; or structural, such as those which describe novel biomaterials, bone and cartilage substitutes. In terms of the organs or tissues targeted by many of the patents, bone and cartilage stand out, in keeping with the advanced state of research for these tissues as compared to other more complicated organ systems, such as the kidney, lung, or heart, which will require considerably more research and development before patents and supporting methodologies are seen. Finally, CHI found that of the 100 or so patents examined, nineteen were declared by the inventors to have resulted fully or partially from a government grant. Of these, NIH had eight, NASA six, NSF three, and DHEW116 two.
|