|
The Emergence of Tissue Engineering as a Research Field |
|
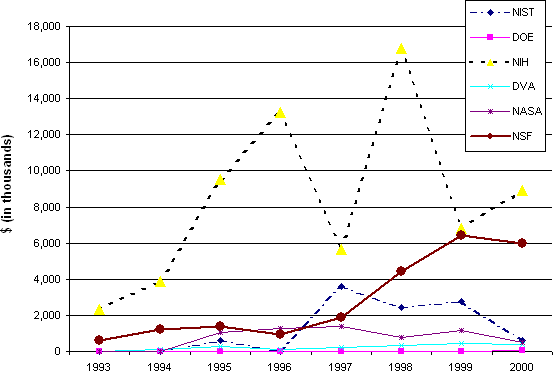
Back to Table of Contents View PDF of this document (69 Kb) 7.0 Institutional Support of Tissue EngineeringIn addition to the individual researchers in academia and industry, key players in a field can include the federal agencies that provide financial support, professional societies and other institutions designed to promote and advance a field's mission. Professional societies may promote educational programs, research applications, and the development of professional standards in an overall effort to advance the dissemination of knowledge in a particular field or research area. As networking facilitators, conferences, meetings, and other symposia supported by societies may be instrumental in bringing together researchers who work on similar topics but may not otherwise collaborate. Several groups have emerged in tissue engineering's development. The National Science Foundation is one of many organizations - including the U.S. Federal government and foreign governments, industry and non-profit foundations - that have funded tissue engineering research in the United States and around the world. The WTEC report estimates that nearly $3.5 billion dollars have been spent on tissue engineering in the last decade. Of this, less than 10% has come from the U.S. Federal government117. According to their definition, the National Science Foundation, primarily through its Directorate for Engineering, provided less than 1% of worldwide support for tissue engineering, but nearly 7% of U.S. Federal government support. Table 7.1 below and the accompanying Figure 7.1 examine U.S. Federal government funding of tissue engineering in greater detail, using a more restrictive definition. They provide a partial glimpse into Federal support of TE using data from RaDiUS, a database that tracks the research and development activities and resources of the Federal government.118 Of all Federal agencies supporting biomedical science and engineering, NSF is second only to NIH in providing financial support to TE, and has increased both its level and share of Federal funding in the past few years. This chapter summarizes the support of the field by the key federal agencies involved: National Institutes of Health (NIH), National Institute of Standards and Technology (NIST), National Aeronautics and Space Administration (NASA), Food and Drug Administration (FDA), and finally, NSF. Before turning to NSF's role in supporting tissue engineering, we provide brief descriptions of major efforts of other Federal agencies that have been active in tissue engineering in different ways. Table 7.1 Federal Awards for Tissue Engineering Research 1993 - 2000, by Agency (000 $)*
* While RaDiUS did not pull any records from DOD, we are aware of DARPA's role in supporting activities related to Tissue Engineering. In the last five years, DARPA has funded research in cell-based biosensors. Figure 7.1 Federal Awards for Tissue Engineering Research, by Year and by Agency (1993 - 2000) * Source: RaDiUS database, grants containing term "tissue engineering" 7.1 National Institutes of Health (NIH)Support of tissue engineering from the NIH has come in several forms over the years. While an official program/focus area in tissue engineering did not begin until the late 1990's, many key researchers in the field attribute much of the early support they received as coming from the NIH. Traditionally known for its support of fundamental research, NIH has supported many of the basic science advancements, which led to tissue engineering as we know it today. Much of the past work done on cell culture methods, for example, was supported by NIH funding. Nonetheless, NIH was a relative newcomer to tissue engineering per se. In a 1998 article in Science, for example, researchers berated NIH's lack of interest in the field:
In more recent years, NIH's focus on TE has increased dramatically:120
In 2000, the NIH launched its newest institute, the National Institute for Biomedical Imaging and Bioengineering (NIBIB), which aims to "�improve health by promoting fundamental discoveries, design and development, and translation and assessment of technological capabilities in biomedical imaging and bioengineering, enabled by relevant areas of information science, physics, chemistry, mathematics, materials science, and computer sciences121." 7.2 National Institute for Standards and Technology (NIST)NIST's Advanced Technology Program (ATP) began in 1988 and was designed to focus on commercial applications of high-risk cutting edge basic scientific research, such as biology, a field NIST did not yet participate in. The first ATP competition was held in 1990-91 and the first TE award was made in the 2nd round of competition to Acerm Biosciences, a company growing stems cells from bone marrow. In the years subsequent, NIST added projects at the rate of 1-2 per year on topics such as building better scaffolding, bioreactors, and xenotransplantation.122 Serious efforts in tissue engineering, however, did not occur until the mid-late 1990s. In 1993-94, a two day conference was held on cutting-edge biotechnology and involved individuals from several agencies including Francis Collins of NIST, Fred Heineken of NSF, and Kiki Hellman of FDA. The group identified 'hot topics' in a hierarchy-tissue engineering being one of those near the top of the list. For each, a set of key issues was identified and listed: in what areas is there enough basic science research and where is there a black hole? Other technologies identified at this meeting included DNA diagnostics, TE, vaccines, gene therapy, and biosensors. Approaches for the development of specific industries was discussed. Stan Abramowitz, who headed the early NIST/ATP efforts was heavily involved in this. Staff attended conferences to "find the right people" to get advice, and to head these initiatives. As a follow-up to these efforts, a series of white papers were solicited in 1995 in preparation for a focused competition in TE. The first focused program was initiated in 1997 when 56 proposals were submitted and 12 awarded (for a total of $12 million that year). Focused competitions in tissue engineering ended for ATP in 1998, however. The ATP Program has been instrumental in funding high-risk commercial applications of ideas that venture capitalists are unlikely to support-typically these are multidisciplinary projects which require academic collaboration. Dr. Rosemarie Hunziker, a former Program Officer at ATP, believes that NIST/ATP support of TE gave it credibility, an important action, especially in light of its heavy private sector orientation123. 7.3 National Aeronautics and Space Administration (NASA)Beginning in the late 1970's, NASA's efforts in tissue engineering have focused on development of techniques for three-dimensional cell and tissue culture. The agency's interest in tissue engineering comes from a need to understand how cells and tissue behave in space. Laboratory cultures which mimic live human tissue can be used as models to conduct space research and research on deadly diseases, such as cancer-eliminating the need for human test subjects. In the late 1980's, the work of Milbourne and Wolf of the Johnson Space Center culminated in the development of the rotating bioreactor, a horizontal rotating wall vessel with a center oxygen membrane, which promoted cell and tissue growth under a variety of conditions. A patent on the device was filed in 1988 and issued in 1991124. While initially referred to under the agency's biotechnology heading, NASA officially recognized these efforts as "tissue engineering" when the term appeared for the first time in a 1994 program update. Since then, NASA has continued to fund selective research on bioreactors and related topics and has provided a steady stream of funding to individuals in this area. Much of the work done by Lisa Freed and Gordana Vunjak-Novakovic of MIT has been supported by NASA's Cellular Biotechnology Area. 7.4 Food and Drug Administration (FDA)The FDA has taken an interest in tissue engineering since the early 1990's. In 1990, the Center for Devices and Radiological Health (CDRH) in conjunction with the State of Maryland held a conference to examine biological applications with device use. Among the topics raised was tissue engineering. Dr. Kiki Hellman, head of the CDRH, saw potential in the field and became interested in exploring it further. Two years later, a workshop was held to examine promising new technologies—tissue engineering being one of them. From an agency perspective, the FDA was proactively involved in the technology's development.125 According to Dr. Hellman, the FDA made a concerted effort to deal with in-house needs (staff training, education) so that they could anticipate the technology and be prepared to deal with the any technical or scientific issues that would arise. Many of our interviewees also state that the FDA has been instrumental in allowing products to move to market, encouraging the development of standards to measure devices (in conjunction with the ASTM Committee F04 Medical and Surgical Materials and Devices). In all, many experts in the field laud their efforts, stating that the FDA is not a force standing in the way of progress. 7.5 Other Institutions involved in Tissue EngineeringThe Tissue Engineering Society International (TESI) In 1995, Charles Vacanti started The Tissue Engineering Society (now known as Tissue Engineering Society International or TESI). In its early days, the society served to foster communication between individuals in the tissue engineering world through sponsorship of meetings and conferences. In 1995, the Society published the first issue of Tissue Engineering, the first dedicated journal in the field. Despite the placement of several prominent tissue engineers on its editorial board, the journal lacked popularity. Many researchers continue to preferentially publish in more traditional disciplinary journals or general peer-reviewed publication like Science or Nature. More recently, the society has grown to include a more international focus and serves as a networking organization to foster dialogue between international researchers in the field.126 The Pittsburgh Tissue Engineering Initiative A unique and somewhat unusual contributor to the field of tissue engineering has been the Pittsburgh Tissue Engineering Initiative (PTEI). The organization was founded by Peter Johnson (founded 1994) a former surgeon at the University of Pittsburgh. After becoming chairman of the department of plastic surgery, Johnson was in a unique position to form a network of scientists in the Pittsburgh area. Based largely upon his own interest in tissue engineering and its potential, Johnson pushed to develop a joint technology transfer policy between the major Pittsburgh area universities, and PTEI was born. In its beginnings, PTEI served mostly as a networking mechanism—to raise funds to provide support to TE research in the surrounding area. The initiative had four key components: (1) technology development grants, (2) summer research internships to increase student base in TE, (3) biotech exposure (with a minority focus), and (4) a guest speaker program (a coordinated effort to bring big names in the field to Pittsburgh)127. PTEI raised money and support and led the 1st TE biotech company in the area. Currently, there are at eight firms in the area. PTEI has encouraged an amalgamation and growth of the critical mass necessary to sustain development in TE for the Pittsburgh community. As a result, Pittsburgh is rapidly becoming a center for informatics—a new branch in the field of tissue engineering. PTEI also serves as a model for other cities to advance research and development in TE. Toronto is expanding their TE workforce utilizing the PTEI model. The Whitaker Institute for Biomedical Engineering The mission of The Whitaker Foundation is to promote better human health through advancements in medicine. This is accomplished through a series of competitive grant programs that support research and education in biomedical engineering at academic institutions in the United States and Canada128. Whitaker has been instrumental in funding bioengineering programs around the country. Most notable for TE, they, in 1999, established the Whitaker Institute for Biomedical Engineering (WIBE) at UCSD under the leadership of Y.C. Fung. Much of the fundamental work supported under this institute will contribute to advancements in the field of TE. However, the foundation has been reluctant to classify TE as an independent entity, but rather a sub-field of bioengineering129. Multi-Agency Tissue Engineering Sciences (MATES) Working Group More recently, many agencies of the federal government have become interested in supporting the mission of tissue engineering. In an effort to coordinate this support, representatives from each of several federal agencies came together in 2000, under the leadership of Drs. Fred Heineken (NSF) and Kiki Hellman (FDA) to form the Multi-Agency Tissue Engineering Science (MATES) Working Group. MATES has three major goals: (1) to facilitate communication (and prevent redundancy of funding) across departments/agencies by regular information exchanges and a common web site, and (2) enhance cooperation through co-sponsorship of scientific meetings and workshops, and facilitate the development of standards, and (3) to monitor technology by undertaking cooperative assessments of the status of the field130. MATES has developed a web-site which they hope will become "one-stop-shopping" for those seeking information on the field (such as information about federal funding, scientific meetings, regulatory guidance and standards development). Participating agencies in MATES include the Department of Commerce (NIST), the Department of Energy (DOE), the Department of Defense (DARPA), the Department of Health and Human Services (FDA, NIH), the National Aeronautics and Space Administration (NASA), and the National Science Foundation (NSF). The panel, co-chaired by Drs. Frederick Heineken of NSF and Kiki Hellman of the FDA also focuses on some regulatory, legal, and ethical issues for TE. A particular area of interest has been a global regulatory initiative for TE—an effort that will require the development of standards for testing tissue engineered products. A major contribution of the MATES group is the recent World Technology Evaluation Center (WTEC) study on tissue engineering, which attempts to summarize the technical contributions of current work in TE and to estimate the funding for TE from each of the federal agencies131. The study has become a focal point for activities of the working group, under the auspices of the Subcommittee on Biotechnology, Committee on Science of the President's National Science and Technology Council (NSTC). The results of the WTEC study were used by MATES to plan a joint interagency program announcement in tissue engineering issued under the new National Institute for Biomedical Imaging and Bioengineering, which solicits research aimed at addressing specific gaps in tissue engineering.
|