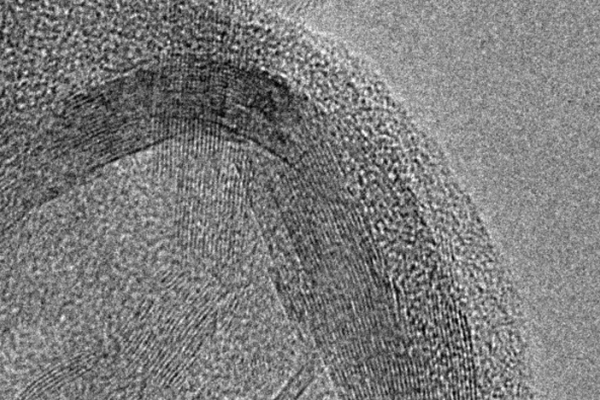
We support breakthroughs in chemistry that enhance nearly every aspect of daily life, from pharmaceuticals to plastics, environmental cleanup to battling pandemics.
NSF-backed research has led to new ways to identify infection gateways of the coronavirus, made an aerogel out of common egg whites that can filter water and store energy, and devised methods to break down or reuse plastic waste.
What we support

Discovery and innovation in chemistry
We support research on the composition, structure, property and reactivity of the basic components of matter.

World-class research centers and facilities
We support a network of research centers and user facilities that tackle grand scientific and societal challenges and train the next generation of chemists.

Education and workforce development
We support training and educational opportunities that allow more people and communities to explore a rewarding career in the chemical sciences.
Sustainable chemistry
We support chemical research aimed at improving the sustainability of resources such energy, materials and water.
Facilities
We support facilities housing advanced chemical research instrumentation and capabilities that scientists across the country are able to use in their work.
NSF Chemistry and Materials Center for Advanced Radiation Sources
ChemMatCARS provides instruments and experiment stations that use the high-energy X-rays provided by the Advanced Photon Source at the Department of Energy's Argonne National Laboratory.
NSF Ion Cyclotron Resonance Facility
The ICR facility at the NSF National High Magnetic Field Laboratory houses four Fourier transform ion cyclotron resonance mass spectrometers that enable fast and high-resolution analysis of complex chemical mixtures.
NSF National Extreme Ultrafast Science
NeXUS provides researchers with access to a high-speed pulse laser enabling their investigation of phenomena such as the ultra-fast motion inside molecules.
Featured news
Educational resources
View lesson plans, activities and multimedia for K–12 audiences that focus on explorations of chemistry.
View the resources