What are polymers?
Polymers are large molecules made by linking many smaller units together, like beads on a string. They occur in both natural and synthetic materials, including rubber, silk and everyday plastics.
These long chains are customizable and can be flexible or rigid, soft or tough — making them suitable for use in everything from food packaging and clothing to medical devices and electronics.
Examples of polymers:
Rubber — Tires, surgical gloves and waterproof products.
Silk — Clothing, sutures and specialty textiles.

Credit: SPT Paul Topham, CC BY-SA 4.0, via Wikimedia Commons CC BY-SA 4.0, via Wikimedia Commons
Laying the groundwork
Pioneering NSF-funded research laid the foundation for modern polymer science, enabling large-scale production of durable plastics, rubbers and synthetic fibers that transformed daily life and strengthened U.S. manufacturing.

Conducting electricity
In the 1970s, NSF-supported research led to the discovery of conductive polymers, a new class of plastics capable of carrying an electric charge.
This breakthrough opened the door to flexible electronics, solar panels, biosensors and more, ultimately earning a share of the 2000 Nobel Prize in chemistry.

Restoring blood flow
Angioplasty was first developed in the late 1960s to open clogged arteries, but early procedures often resulted in re-narrowing. NSF-supported research beginning in the 1980s led to the development of a biostable elastomeric (rubbery) polymer that allowed stents to hold pharmaceutical products when coated with it.
This coating prevents inflammation and delivers time-released medications to prevent reclogging, significantly decreasing the need for open-heart surgeries.
Rewriting the rules of polymer production
In the 1990s, NSF-supported research helped advance a powerful chemical reaction called metathesis, which allows scientists to break and reform carbon double bonds in a controlled way to build new molecules.
This breakthrough streamlined polymer creation — reducing energy use, limiting hazardous byproducts and lowering production costs. It enabled large-scale manufacturing of new plastics and advanced materials now widely used in automotive parts, electronics, coatings and pharmaceuticals.
The work earned Robert Grubbs and Richard Schrock a share of the 2005 Nobel Prize in chemistry.
Polymers reimagined
NSF-funded researchers are pushing the boundaries of what is possible in materials science, creating polymers with novel properties that unlock entirely new applications, such as:

Shape-memory polymers
NSF-supported researchers developed shape memory polymers — materials that undergo reversible shape transformations in response to heat and magnetism or electricity — enabling applications in smart textiles and medical implants that conform to the body.
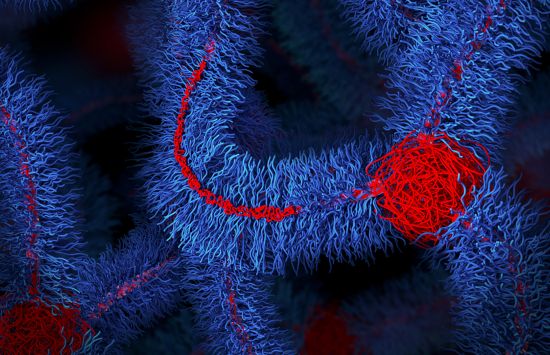
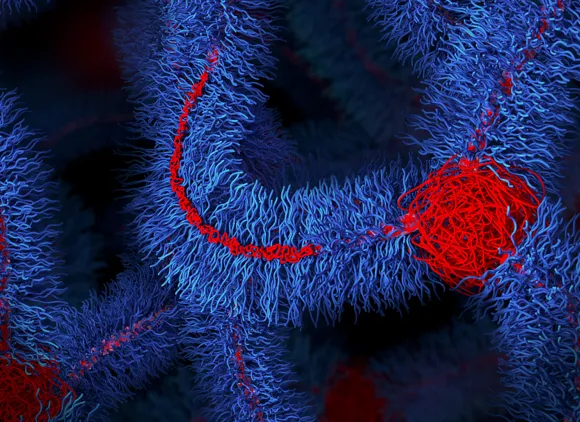
The "impossible" polymer
With NSF support, researchers developed an "impossible" polymer that is both stiff and stretchable, overcoming a centuries-old materials trade-off.
The material's bottlebrush architecture is now being explored for durable heart implants, prosthetics, soft robotic systems and other electromechanical devices that must bend without breaking.
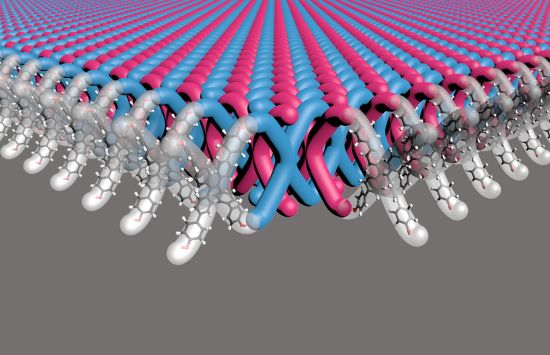
Chainmail polymer
NSF-funded scientists developed the first "chainmail" polymer, a flexible, 2D mechanically interlocked material as strong as armor, offering potential uses in protective gear for first responders and military personnel.

Ultra-strong composites
NSF-funded scientists engineered ultra-light, ultra-strong polymer nanocomposites, including nanoscale 3D-printed materials that absorb twice as much energy as comparable materials.
These lightweight, impact-resistant structures offer improved protection for satellites, drones, aircraft and microelectronics while supporting more efficient systems.

AI-designed polymers
Artificial intelligence algorithms developed by NSF-funded scientists could revolutionize how scientists and manufacturers identify and develop polymers with precise molecular architectures and properties.
By analyzing millions of polymer structures and learning the "chemical language" that governs how atoms connect, these tools can rapidly screen vast numbers of possible materials to predict which combinations will produce polymers with desired characteristics such as flexibility, water resistance or electrical conductivity.

Polymers can quantum too
With NSF support and leadership, researchers are using self-assembled polymers to build quantum materials and devices with precisely controlled nanoscale structures.
By combining polymers with metals and ions, scientists are creating hybrid materials with tunable quantum properties, such as superconducting nanowires and quantum optical systems, that could enable new generations of quantum and microelectronic technologies.

Building the infrastructure for next-generation polymers
Through programs such as the NSF Centers for Chemical Innovation and the NSF Materials Research Science and Engineering Centers, NSF is driving advances in polymer design and advanced materials and moving cutting-edge materials discoveries from the laboratory to real-world applications.
For example, researchers are developing polymers that can self-replicate, adapt and strengthen under stress, improving performance and resilience in applications ranging from infrastructure to medicine. Others are developing ways to control and reconfigure materials in real time that could revolutionize industries such as advanced manufacturing, biotechnology and robotics.